A potential link exists between the tumor microenvironment and pathological phenotypes of the peripheral nervous system in vivo, supposedly driving pain states in cancer-induced bone pain patients (CIBP). We aim to recapitulate this phenomenon in a controlled high-throughput in vitro setting with the goal of studying primarily cancer-neuron interactions and the effects on neuronal excitability.
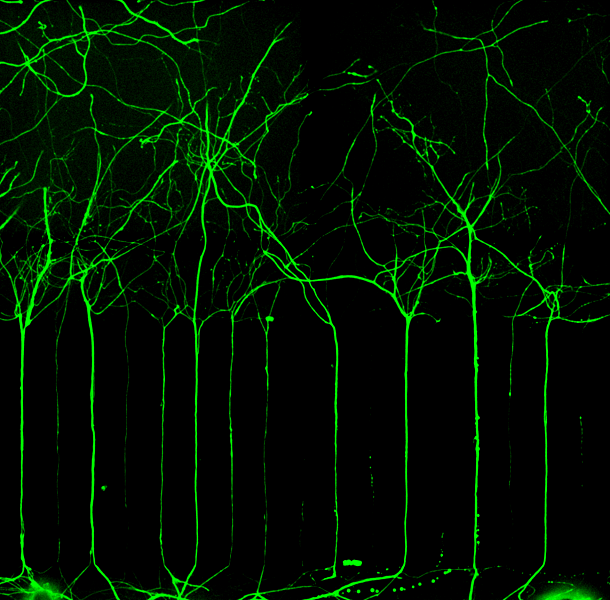
We develop a microelectrode array (MEA) integrated chip to record electrophysiological activity from three-dimensional (3D) culture of sensory neurons innervating tumor-like constructs. The CIBP-on-a-chip system enables carrying out both immunohistochemistry and electrophysiology on single experiments in a multiplexed format – which is a necessity when setting out to link morphological/molecular interactions to alterations in neuronal function. This approach will facilitate the accurate study of bone pain phenotypes in vitro and provide a platform for further discovery of pain-mitigating therapeutic compounds.
Furthermore, as the platform developed towards modeling pain phenotypes, we seized the opportunity to explore its modality-agnostic aspects – and designed its features in a way that potentially allows for innervation of any choice tissue (autonomic/somatosensory).
Project – BONEPAIN II
Funding: The European Union Horizon 2020 Framework Programme for Research and Innovation under the Marie Sklodowska-Curie Grant Agreement No. 814244 (BONEPAIN II). ; This work received financial support from the State Ministry of Baden-Wuerttemberg for Economic Affairs, Labour and Tourism.;